Water is more than just a nuisance for diesel engines, it’s a destructive force that can compromise your entire fuel system. While diesel will always contain some water, the goal is to keep levels low enough to avoid serious problems.
Understanding how water enters your fuel, how it causes damage, and how it's best managed is essential for the protection of your equipment and operations.
Why Water is a Problem in Diesel Fuel
Modern diesel engines have no tolerance for handling free water. Here’s how water harms your system:
Direct Damage
Water can destroy both fuel tanks and engine parts by causing:
- Rust: Iron and steel surfaces oxidize in contact with water, like other hard particulates; rust will cause abrasive wear to parts.
- Corrosion: Corrosion is one of the most common causes of injector problems. Water combines with acids in the fuel to corrode both ferrous and non-ferrous metals. This is made worse when abrasion exposes fresh metal surfaces that readily corrode.
- Abrasion: Water has lower viscosity than diesel, therefore providing less of a lubricating "cushion" between the opposing surfaces of moving parts. This leads to increased abrasion.
- Etching: Etching is caused by water-induced fuel degradation, which produces hydrogen sulfide and sulfuric acid that "eat" metal surfaces.
- Pitting and Cavitation: Pitting is caused by free water flashing on hot metal surfaces. Cavitation is caused by vapor bubbles rapidly contracting (imploding) when exposed to sudden high pressure, which causes them to condense back into a liquid. These water droplets impact a small area with great force, causing surface fatigue and erosion.
- Spalling: Occurs due to hydrogen embrittlement and pressure. Water is forced into microscopic cracks in metal surfaces. Then, under extreme pressure, it decomposes and releases hydrogen in a micro-explosion, which enlarges the cracks and creates wear particles.
- Ice Crystals: Free water in fuel can freeze, creating ice crystals that behave just like any other hard particulate, creating wear in fuel systems and (in large volumes) clogging fuel filters.

Indirect Damage
Water doesn’t stop at physical damage. It also accelerates chemical breakdown:
- Soft Solids: Water is polar. Certain chemicals in additives are polar. Hydrocarbons are non-polar. This means that water and polar chemicals are attracted to each other. In the presence of free water, the chemical molecules will sometimes disassociate themselves from the hydrocarbon chain of the additive and combine with water molecules to form a new substance. The new material is a soft solid that precipitates out of the fuel and can rapidly clog filters or create engine deposits. See additive stability for more information.
- Microbial Growth: Like most living organisms, bacteria and fungi need both food and water to survive. If free water is present, microbial growth can proliferate, creating slimes that foul your fuel and acids that corrode your tank and fuel system.
- Fuel Oxidation: Free water accelerates the oxidation process and encourages the formation of acids, gum, and sediments known generally as fuel degradation products.

How Water Gets into Diesel
Water contamination can occur at many points in your fuel handling process:
- Fuel delivery – Fuel may already contain water from upstream sources.
- Free water drop-out – fuel is wetter than the saturation point.
- Condensation – Thermal cycling causes moisture to form inside tanks.
- Leaks – Rain, pressure washing, or groundwater can infiltrate tanks.
- Atmospheric absorption – Humid air can transfer moisture into diesel.
- Human error – Poorly sealed vents, caps, or ports invite water ingress.
Forms of Water in Diesel
Water in diesel exists in three forms:
- Dissolved Water – Invisible and usually harmless below the saturation point.
- Free Water – Droplets that separate from fuel and settle to the bottom.
- Emulsified Water – Tiny suspended droplets that remain evenly mixed in fuel.
Warm fuel can hold more water than cool fuel. Problems occur once the fuel’s water content exceeds the saturation point. As fuel cools overnight, the same fuel can hold less water than it did when warm, and free water can drop out every day.
How Much Water is Too Much?
While zero water would be ideal, it’s not realistic. The key is keeping water levels below saturation to prevent separation into free water.
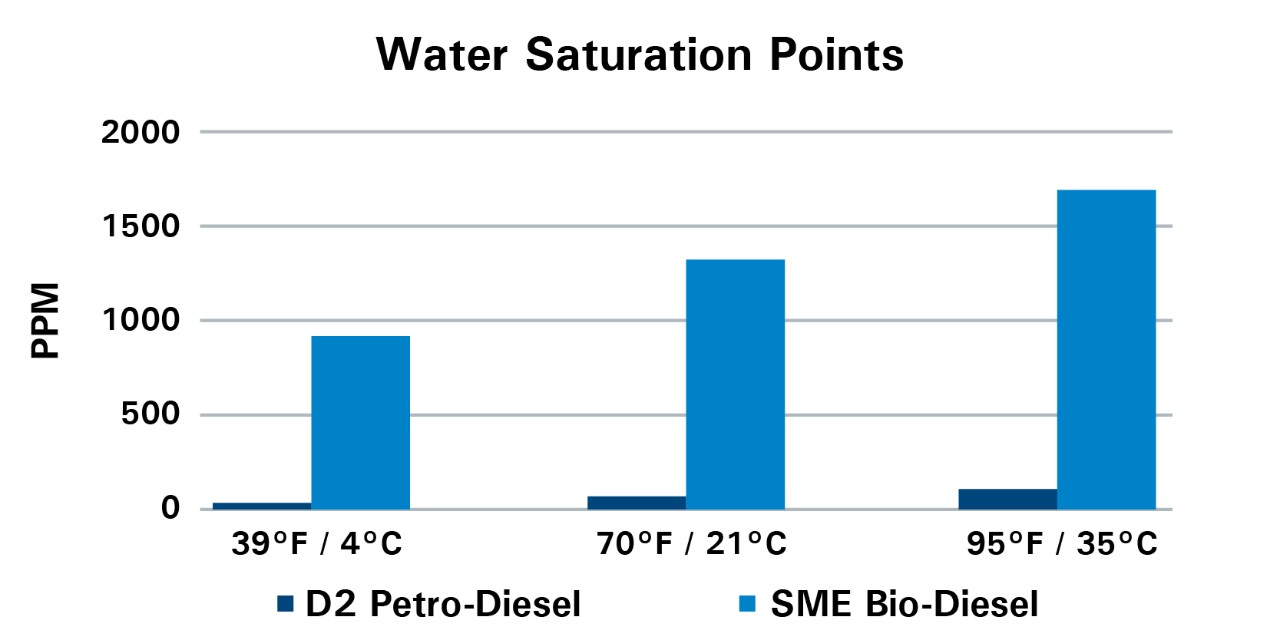
- Saturation levels range from 50 to 1800 ppm, depending on temperature and biodiesel content
- Biodiesel holds more water than petro-diesel, but blending the two doesn’t result in a simple average; free water may form as a result of the blending process.
How to Measure Water in Diesel
Several methods exist, each with its own pros and cons:
- Tank dip tests – Use water-detecting paste on a stick to check for bottom water. Simple and fast, but limited to free water.
- Inline sensors – Provide real-time data on dissolved water content as a percentage of relative humidity.
- Karl Fischer titration – A highly accurate lab test detecting water down to about 50 ppm.
How to Prevent Water Contamination
To stop water before it causes damage:
- Control fuel deliveries:
- Be delivered first, you don’t want the settled water and dirt from the tank bottoms
- Install water blocking filters at your bulk tank inlet.
- Protect from the environment:
- Seal access covers and install deliquescent breathers.
- Shield tanks from temperature swings to minimize condensation.
- Avoid ground-level fill points that can let rainwater in.
- Manage storage conditions:
- Rotate fuel regularly.
- Keep tanks topped off to reduce air (and moisture) exposure.
- Monitor water levels and routinely remove free water.
The Trouble with Removing Water
Removing water is more complicated with today’s ultra-low sulfur diesel (ULSD). Here’s why:
- ULSD contains surfactant additives that interfere with coalescing filters.
- These surfactants make traditional water separators much less effective at high flow rates.
- Water blocking medias are often required to ensure dispensed fuel is dry.
- Draining free water from tanks is the most cost-effective method for larger volumes of free water.

The Bottom Line
Water is unavoidable in diesel fuel, but damage is not. With proper testing, smart tank design, and the right combination of filtration and storage strategies, you can protect your engines from water-related failures.
Investing in proactive fuel water management helps you avoid costly repairs, unexpected downtime, and long-term performance issues.

